Charge It!!!
Everything in the world from Apples to Zugats are made up of tiny little particles called atoms. They are so small that they cannot even be seen with a microscope!
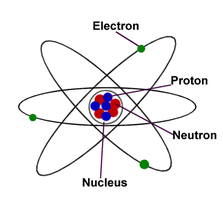
Nucleus: The center of the atom. Tiny particles with positive charges are found inside the nucleus
Electron: Tiny particles that float around the nucleus and are negatively charged.
Imbalance of Charges: When the number of negative charges and positive charges in an atom are not equal
Electron: Tiny particles that float around the nucleus and are negatively charged.
Imbalance of Charges: When the number of negative charges and positive charges in an atom are not equal
Negatively Charged Atom: If there are more negative charges than positive ones we say the atom is negatively charged

Positively Charged Atom: If there are more positive charges than negative ones we say the atom is positively charged.
Neutral Atom: If there are the same amount of positive and negative charges, we say that atom is uncharged or neutral
Electrically Charged: When an atom is either positively or negatively charged
-------------------------------------------------------------------------------------------------------------------------------------------------------------------------------
Neutral Atom: If there are the same amount of positive and negative charges, we say that atom is uncharged or neutral
Electrically Charged: When an atom is either positively or negatively charged
-------------------------------------------------------------------------------------------------------------------------------------------------------------------------------
But How!?!?!

Atoms generally start out balanced. But when some atoms get close to one another, one may pick up an electron from the other one. The atom that gets the electron is now negatively charged and the atom that lost the electron is now positively charged.
(Image: FBcoversking.com)
Electricity:The Movement of Electrons
(Image: FBcoversking.com)
Electricity:The Movement of Electrons
